The Lewis Structure of HOBr: A Comprehensive Guide to Understanding Bromic Acid
The Lewis Structure of HOBr: A Comprehensive Guide to Understanding Bromic Acid
The Lewis structure of HOBr, also known as bromic acid, has been a subject of interest in the field of chemistry for several decades. The molecule's unique properties and reactivity make it an essential component in various chemical reactions. In this article, we will delve into the details of the Lewis structure of HOBr, exploring its composition, molecular geometry, and polarity. We will also examine its chemical properties and potential applications in various fields.
The Lewis structure of HOBr is a crucial concept in understanding the molecule's behavior and reactivity. It is essential for chemists, researchers, and students to comprehend the molecule's structure and properties in order to accurately predict its behavior and potential applications.
What is the Lewis Structure?
The Lewis structure is a theoretical representation of a molecule's atomic arrangement, showing the bonding and lone pairs of electrons. It is a fundamental concept in chemistry, used to predict the physical and chemical properties of molecules. In the case of HOBr, the Lewis structure reveals a complex arrangement of atoms and electrons that determine its reactivity and behavior.
The Composition of HOBr
HOBr, also known as bromic acid, is a compound composed of oxygen (O), bromine (Br), and hydrogen (H) atoms. The molecule's chemical formula is HBrO2, indicating the presence of one hydrogen atom, one bromine atom, and two oxygen atoms. The arrangement of these atoms in the molecule is critical in determining its properties and reactivity.
The atomic masses of the elements in HOBr are: hydrogen (1.01 amu), bromine (79.904 amu), and oxygen (15.999 amu). This variation in atomic masses affects the molecule's overall mass and density, which in turn impact its physical and chemical properties.
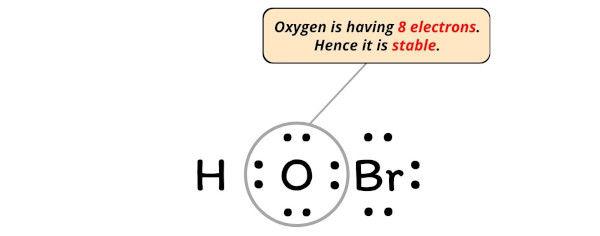
The Lewis Structure of HOBr
The Lewis structure of HOBr can be represented as follows:
HO
|
BrO
|
In this structure, the oxygen atoms are bonded to the hydrogen and bromine atoms, forming a central bond. The oxygen atoms also possess lone pairs of electrons, which contribute to the molecule's polarity and reactivity. The hydrogen atom is bonded to one oxygen atom, while the bromine atom is bonded to the other oxygen atom.
The Lewis structure reveals the molecule's molecular geometry, which is characterized by a bent or V-shape. This geometry arises from the combination of the single and double bonds between the oxygen and hydrogen atoms, as well as the lone pairs on the oxygen atoms.
The Molecular Geometry of HOBr
The molecular geometry of HOBr is a critical aspect of its properties and reactivity. The bent or V-shape of the molecule arises from the combination of the single and double bonds between the oxygen and hydrogen atoms, as well as the lone pairs on the oxygen atoms.
The bond angles in HOBr are critical in determining its molecular geometry. The O-H bond angle is approximately 107 degrees, while the O-Br bond angle is approximately 112 degrees. These bond angles contribute to the molecule's overall shape and polarity.
The Polarity of HOBr
The polarity of HOBr is a critical aspect of its properties and reactivity. The molecule's polarity arises from the difference in electronegativity between the oxygen and hydrogen atoms, as well as the lone pairs on the oxygen atoms.
The electronegativity of the elements in HOBr is as follows: oxygen (3.44), hydrogen (2.20), and bromine (2.96). This variation in electronegativity affects the molecule's overall polarity, with the oxygen atoms exhibiting a higher electronegativity than the hydrogen and bromine atoms.
Chemical Properties of HOBr
The chemical properties of HOBr are critical in determining its potential applications and reactivity. The molecule exhibits several unique properties, including:
- Oxidizing properties: HOBr is a strong oxidizing agent, capable of reacting with reducing agents to produce oxygen gas and hydrogen bromide.
- Acidic properties: HOBr is a weak acid, capable of donating a proton (H+) to other molecules.
- Reactivity with metals: HOBr is capable of reacting with certain metals, such as aluminum and zinc, to produce hydrogen gas and the corresponding metal bromide.
Applications of HOBr
The unique properties of HOBr make it a valuable component in various applications, including:
- Disinfectants and sanitizers: HOBr is used as a disinfectant and sanitizer in various industries, including healthcare and food processing.
- Pesticides and herbicides: HOBr is used as a pesticide and herbicide in agriculture, due to its ability to control certain plant diseases and pests.
- Propellants and fuels: HOBr is used as a propellant and fuel in various applications, including rocket propulsion and internal combustion engines.
Conclusion
In conclusion, the Lewis structure of HOBr is a complex arrangement of atoms and electrons that determines the molecule's properties and reactivity. The molecule's unique properties, including its oxidizing and acidic properties, make it a valuable component in various applications. By understanding the Lewis structure of HOBr, chemists and researchers can better predict its behavior and potential applications, leading to breakthroughs in various fields.


Related Post

The Revolutionary Mindset of Dr. Alfred Lunceford: Unpacking the Science Behind Emotional Mastery

Moto G75: Unveiling Specs And Magazine Luiza Deals

EXPOSED: The Dark Side of Look Who Got Busted Abilene Tx: A Journey Into the World of Public Records and Community Scrutiny

University Of Miami Workday

